
Table of Contents
- What is the definition of atom
- Probing the atom
- Major sub atomic particles
- The Bohr model of atom
- Conclusion
What is the definition of atom
An atom is the basic unit of matter, composed of a nucleus of positively charged protons and neutral neutrons, surrounded by a cloud of negatively charged electrons. Atoms are incredibly small and cannot be seen with the naked eye. They are the building blocks of all matter and are classified based on the number of protons in the nucleus, which determines their chemical properties and place in the periodic table. Atoms can combine to form molecules, which in turn make up all of the substances in the world around us.
Probing the atom
Since the dawn of civilization, humans have been fascinated by the natural world and the laws that govern it. With the advent of modern science, our understanding of the world has grown by leaps and bounds, and nowhere is this more evident than in the field of atomic physics. By probing the atom, scientists have uncovered the fundamental nature of matter, and in doing so, have unlocked some of the greatest mysteries of the universe.
At its core, the atom is a tiny, sub-microscopic particle that serves as the building block of matter. Each atom is made up of a nucleus, which contains positively charged protons and neutral neutrons, surrounded by a cloud of negatively charged electrons. Understanding the behavior of these particles is essential to understanding the nature of the physical world, and scientists have employed a variety of tools and techniques to probe the atom in order to uncover its secrets.
One of the most important tools in atomic physics is the particle accelerator. These machines use powerful magnetic fields to accelerate particles to incredibly high speeds, allowing scientists to study the behavior of particles at the atomic level. By colliding these particles with other particles or with solid objects, researchers can observe the results of these interactions and gain insight into the fundamental nature of matter.
Another important technique for probing the atom is spectroscopy. This involves analyzing the way in which atoms absorb or emit light at different wavelengths. By studying the patterns of absorption or emission, scientists can determine the energy levels of the electrons in the atom, as well as other important properties such as the atom’s mass and the strength of the forces between the particles.
In addition to these techniques, scientists also use a variety of other tools and methods to probe the atom, including X-ray crystallography, electron microscopy, and quantum mechanics. Each of these approaches provides a unique perspective on the behavior of particles at the atomic level, and together they have enabled researchers to build a comprehensive understanding of the nature of matter.
The first of these smaller particles was discovered by British physicist James (J. J.) Thomson in 1897. Named the electron, this particle is negatively charged. (It is the flow of these particles that produces currents of electricity, whether in lightning bolts or in the wires leading to your lamp.) Because an atom in its normal state is electrically neutral, each electron in an atom must be balanced by the same amount of positive charge.
The next step was to determine where in the atom the positive and negative charges are located. In 1911, British physicist Ernest Rutherford devised an experiment that provided part of the answer to this question. He bombarded an extremely thin piece of gold foil, only about 400 atoms thick, with a beam of alpha particles (Figure 1). Alpha particles (α particles) are helium atoms that have lost their electrons and thus are positively charged. Most of these particles passed though the gold foil just as if it and the atoms in it were nearly empty space. About 1 in 8000 of the alpha particles, however, completely reversed direction and bounced backward from the foil. Rutherford wrote, “It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.”
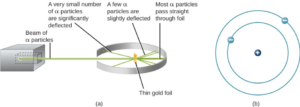
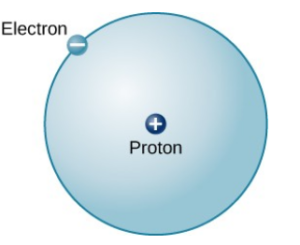
The only way to account for the particles that reversed direction when they hit the gold foil was to assume that nearly all of the mass, as well as all of the positive charge in each individual gold atom, is concentrated in a tiny center or nucleus. When a positively charged alpha particle strikes a nucleus, it reverses direction, much as a cue ball reverses direction when it strikes another billiard ball. Rutherford’s model placed the other type of charge—the negative electrons—in orbit around this nucleus.
Rutherford’s model required that the electrons be in motion. Positive and negative charges attract each other, so stationary electrons would fall into the positive nucleus. Also, because both the electrons and the nucleus are extremely small, most of the atom is empty, which is why nearly all of Rutherford’s particles were able to pass right through the gold foil without colliding with anything. Rutherford’s model was a very successful explanation of the experiments he conducted, although eventually scientists would discover that even the nucleus itself has structure.
The insights gained through atomic physics have had a profound impact on our understanding of the universe. From the development of new materials and technologies to the exploration of the cosmos, the knowledge gained through atomic physics has transformed our world in countless ways. And as we continue to probe the atom and push the limits of our understanding, we can only imagine what new discoveries lie ahead.
In conclusion, probing the atom is a crucial tool for understanding the fundamental nature of matter. By employing a variety of techniques and tools, scientists have uncovered a wealth of knowledge about the subatomic world, and in doing so, have transformed our understanding of the universe. As we continue to explore the mysteries of the atom, we can look forward to even more exciting discoveries and a deeper understanding of the world around us.
Major sub atomic particles
The study of subatomic particles has been an essential field of physics for the past century. At the most basic level, all matter is composed of subatomic particles. The three major subatomic particles that make up atoms are protons, neutrons, and electrons. Each of these particles plays a critical role in the behavior of atoms and the physical world around us.
Protons
Protons are positively charged particles found in the nucleus of an atom. They are relatively heavy, with a mass of approximately 1 atomic mass unit. Protons are essential to the identity of an atom. The number of protons in the nucleus determines the atomic number, which is a unique identifier for each element on the periodic table. The positively charged protons in the nucleus are held together by the strong nuclear force, which is stronger than the electromagnetic force, and is responsible for binding the nucleus of the atom together.
Neutrons
Neutrons are neutral particles found in the nucleus of an atom. They are also relatively heavy, with a mass of approximately 1 atomic mass unit. Neutrons play an important role in determining the stability of an atom. The strong nuclear force is responsible for binding neutrons and protons together in the nucleus. If the number of protons in the nucleus is greater than the number of neutrons, the atom may be unstable and decay into a different element.
Electrons
Electrons are negatively charged particles that orbit the nucleus of an atom. They are much smaller than protons and neutrons and have a negligible mass. Electrons are responsible for the chemical behavior of an atom. The number of electrons in an atom determines its chemical properties, such as its reactivity and ability to form bonds with other atoms. Electrons are attracted to the positively charged nucleus by the electromagnetic force, which is responsible for holding the atom together.
In addition to these three major subatomic particles, there are other subatomic particles, such as muons, gluons, and quarks, that have been discovered through particle accelerators and other high-energy experiments. These particles play important roles in the behavior of subatomic particles, but their impact on the larger physical world is still being explored.
In conclusion, the three major subatomic particles – protons, neutrons, and electrons – are essential components of atoms and play critical roles in determining the behavior of matter. Understanding the nature of these particles and their interactions is essential to our understanding of the physical world around us. As we continue to explore the subatomic realm through new experiments and discoveries, we can look forward to a deeper understanding of the universe and the laws that govern it.
The Bohr model of atom
Rutherford’s model for atoms has one serious problem. Maxwell’s theory of electromagnetic radiation says that when electrons change either speed or the direction of motion, they must emit energy. Orbiting electrons constantly change their direction of motion, so they should emit a constant stream of energy. Applying Maxwell’s theory to Rutherford’s model, all electrons should spiral into the nucleus of the atom as they lose energy, and this collapse should happen very quickly—in about 10–16 seconds.
The Bohr model of the atom is a model proposed by Danish physicist Niels Bohr in 1913 to describe the structure of atoms. At the time, the previous models of the atom had not been able to explain the stability of the atom or the spectra of atomic emission and absorption lines. The Bohr model of the atom provided a new understanding of the structure of the atom and its properties, and paved the way for further developments in atomic physics.
The Bohr model of the atom is based on the idea that electrons orbit the nucleus of an atom in discrete energy levels. According to classical physics, electrons should spiral into the nucleus due to the attraction between the positively charged protons in the nucleus and the negatively charged electrons. However, Bohr proposed that electrons can only occupy certain stable orbits, or energy levels, that are quantized, meaning they have discrete and specific values. Electrons can move between energy levels by absorbing or emitting photons, which have a specific energy that corresponds to the difference in energy levels.
Bohr’s model also proposed that the energy of the electron in an atom is related to its distance from the nucleus. Electrons in higher energy levels are farther from the nucleus than electrons in lower energy levels. The energy required to move an electron from one energy level to another is called the energy difference, and it corresponds to the energy of the absorbed or emitted photon.
The Bohr model was able to explain the spectra of hydrogen atoms, which consist of discrete lines instead of a continuous spectrum. The discrete lines correspond to photons emitted or absorbed when electrons move between energy levels. The model was also able to explain why atoms are stable and do not collapse, as the electrons are restricted to certain energy levels and cannot spiral into the nucleus.
Although the Bohr model provided a breakthrough in atomic physics, it was later shown to have limitations. It could only explain the spectra of hydrogen and hydrogen-like atoms, which have one electron. It did not account for the effects of electron-electron repulsion or for the more complex spectra of atoms with more than one electron. Nonetheless, the Bohr model laid the foundation for the development of quantum mechanics, which provided a more comprehensive understanding of atomic structure and behavior.
The photon and wave perspectives must be equivalent: light is light, no matter how we look at it. Thus, each photon carries a certain amount of energy that is proportional to the frequency (f) of the wave it represents. The value of its energy (E) is given by the formula E=hf
where the constant of proportionality, h, is called Planck’s constant.
The constant is named for Max Planck, the German physicist who was one of the originators of the quantum theory (Figure 5). If metric units are used (that is, if energy is measured in joules and frequency in hertz), then Planck’s constant has the value h = 6.626 × 10–34 joule-seconds (J-s). Higher-energy photons correspond to higher-frequency waves (which have a shorter wavelength); lower-energy photons are waves of lower frequency.
To take a specific example, consider a calcium atom inside the Sun’s atmosphere in which an electron jumps from a lower level to a higher level. To do this, it needs about 5 × 10–19 joules of energy, which it can conveniently obtain by absorbing a passing photon of that energy coming from deeper inside the Sun. This photon is equivalent to a wave of light whose frequency is about 7.5 × 1014 hertz and whose wavelength is about 3.9 × 10–7 meters (393 nanometers), in the deep violet part of the visible light spectrum. Although it may seem strange at first to switch from picturing light as a photon (or energy packet) to picturing it as a wave, such switching has become second nature to astronomers and can be a handy tool for doing calculations about spectra.
Conclusion
Atoms of an element are not all identical and indivisible as Dalton said. They are made of three mains types of particles.
| Particle | mass (amu) | charge |
|---|---|---|
| Electrons | 5.5 x 10–4 | negative (1–) |
| Protons | 1.0073 | positive (1+) |
| Neutrons | 1.0087 | neutral |
The protons and neutrons together form a very small dense center of the atom, called a nucleus. The nucleus contains all the mass of the atom except the small mass of electrons. The electrons move around the nucleus, and occupy a much bigger space than the nucleus, so that most of the atom is empty space.
An element is defined by the number of protons, or atomic number, which is equal to the number of electrons in the neutral element. Atoms of an element can have different numbers of neutrons, resulting in different masses. Isotopes are atoms that have the same number of protons, but different numbers of neutrons. Chemical properties depend mostly on the atomic number, so isotopes are nearly the same chemically. The atomic mass is the average mass of the atom, including all the different isotopes that are likely to be present. If you want to show what isotope of an element you can use special notation, like this: 12C. This is read as carbon-12. The 12 is the mass number, or the number of protons + neutrons. Since carbon always has 6 protons, 12C must have 6 neutrons also.
For More Knowledge Click Here










